Medexus Pharmaceuticals (Medexus) (TSX: MDP) (OTCQX: MEDXF) today announced its operating and financial results and provided a business update for the company’s fourth fiscal quarter and fiscal year ended March 31, 2022. All dollar amounts in this press release are in United States dollars unless specified otherwise.
Financial Highlights
- Delivered total revenue of $20.3 million in fiscal Q4 2022, an increase of 15% compared to fiscal Q4 2021 and, as expected, a decrease of 5% compared to fiscal Q3 2022 partly due to a large order in late fiscal Q3 2022 originally anticipated in fiscal Q4 2022.
- Generated total revenue of $76.7 million in fiscal year 2022, a decrease of 4% compared to fiscal year 2021.
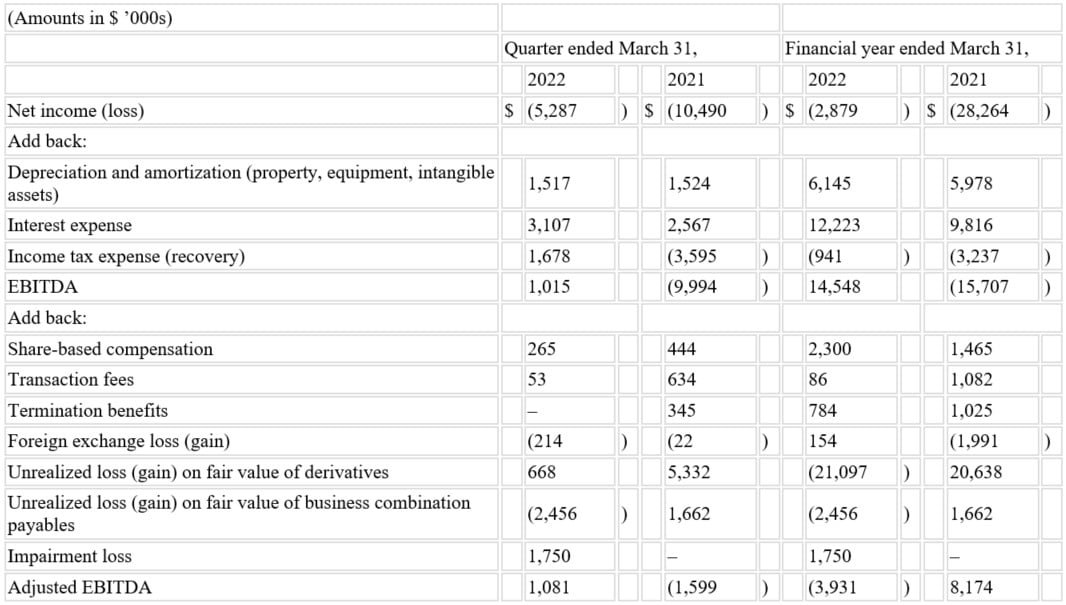
- Achieved adjusted EBITDA* of $1.1 million in fiscal Q4 2022 compared to $(1.6) million in fiscal Q4 2021 and $1.9 million in fiscal Q3 2022.
- Generated adjusted EBITDA* of $(3.9) million in fiscal year 2022 compared to $8.2 million in fiscal year 2021.
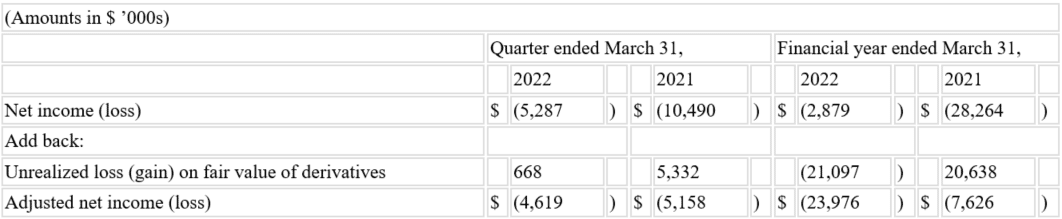
- Produced net loss of $5.3 million in fiscal Q4 2022 and $2.9 million in fiscal year 2022 compared to $10.5 million in fiscal Q4 2021 and $28.3 million in fiscal year 2021.
- Generated adjusted net loss* of $4.6 million in fiscal Q4 2022 and $24.0 million in fiscal year 2022 compared to $5.2 million in fiscal Q4 2021 and $7.6 million in fiscal year 2021.
- Cash and cash equivalents of $10.0 million (with $11.2 million of total available liquidity) at end of fiscal Q4 2022.
* Refer to “Non-GAAP Measures” at the end of this press release for information about adjusted EBITDA and adjusted net income (loss).
Ken d’Entremont, Chief Executive Officer of Medexus, commented, “We are very pleased with the strength and stability we have seen in our base business. We continue to see strong performance in Rasuvo, and Rupall has continued to gain market share and benefit from another strong Canadian allergy season. IXINITY sales have normalized and, with the manufacturing improvements Medexus and our contract manufacturer are undertaking, we expect to realize operational efficiencies going forward that will positively impact our margins down the road.”
Mr d’Entremont continued, “Looking forward at our growth products, the transition for Gleolan in the U.S. is going better than expected and we will begin recognizing product revenue partway through fiscal Q2 and fully in fiscal Q3. We previously estimated that Gleolan had generated US$3 million to US$4 million in revenue in the last full quarter before we licensed the product, and we hope to continue that strong performance following our U.S. relaunch of this product over the coming months. Turning to treosulfan, our partner medac’s collection of data recently requested by the FDA is progressing well, and we are confident we will see an NDA resubmission in July. The review clock for the NDA resubmission, which will run for up to six months, will start after the response is considered complete by the FDA. An FDA approval would then allow a commercial launch of treosulfan in the U.S. in the first half of calendar year 2023.”
Operational Highlights
Operational highlights for the three-month period ended March 31, 2022 and subsequent period include:
- Treosulfan: medac, a strategic partner of Medexus, continues to collect data requested by the FDA in May 2022 to complete the resubmission of medac’s New Drug Application (NDA) for treosulfan. The data collection process is progressing well, and medac continues to expect to respond to the FDA’s information requests in July 2022.
- Gleolan: In March 2022, Medexus acquired the exclusive right to commercialize Gleolan in the United States. Medexus expects to complete the transition of full responsibility for commercializing Gleolan in the United States, and begin recognizing product revenue, in fiscal Q2 2023.
- IXINITY: Unit demand for IXINITY continues to grow, and net sales of IXINITY increased in fiscal Q4 2022 as pharmacy and wholesale customers have now worked through much of their accrued inventory and return to buying patterns better aligned with patient unit demand. Medexus continues to invest in an ongoing initiative to improve the IXINITY manufacturing process. Medexus expects the resulting operational efficiencies to ultimately improve IXINITY gross margins over the coming quarters.
- Rupall: Increasingly severe allergy seasons across Canada and successful sustained execution of sales and marketing initiatives yielded continued strong growth in Rupall sales in fiscal year 2022. This performance continues to position Rupall as one of the fastest-growing antihistamines in the Canadian prescription market. (Source: IQVIA CDH units – Drugstores and hospitals purchases, MAT March 2022.)
Operating and Financial Results Summary for Fiscal Q4 2022
Total revenue reached $20.3 million for fiscal Q4 2022, compared to revenue of $17.6 million for fiscal Q4 2021. The $2.6 million increase is primarily attributable to an increase in net sales of IXINITY during the quarter, and also the continued strong performance of Rasuvo, which is efficiently supported by a moderate allocation of sales personnel, and Rupall.
Operating loss for fiscal Q4 2022 was $2.5 million, compared to an operating loss of $4.6 million for fiscal Q4 2021.
Adjusted EBITDA was $1.1 million for fiscal Q4 2022, compared to $(1.6) million for fiscal Q4 2021. The $2.7 million increase is primarily attributable to the increase in net sales of IXINITY in fiscal Q4 2022 and a $0.9 million expense related to a one-time destruction of IXINITY inventory in fiscal Q4 2021.
Net loss was $5.3 million for fiscal Q4 2022, compared to $10.5 million for fiscal Q4 2021.
Operating and Financial Results Summary for Fiscal Year 2022
Total revenue reached $76.7 million for fiscal year 2022, a decrease of $3.0 million, or 3.8%, compared to fiscal year 2021. The decrease year over year was primarily attributable to a drop in net sales of IXINITY which was partially offset by strong Rupall sales. Unit demand for IXINITY continues to grow, but net sales were lower in fiscal year 2022 as pharmacy and wholesale customers continued to work through inventory on hand for much of the year.
Operating loss for fiscal year 2022 was $15.0 million compared to $1.4 million for fiscal year 2021.
Adjusted EBITDA was $(3.9) million for fiscal year 2022, compared to $8.2 million for fiscal year 2021. The decrease was primarily attributed to significant investments in personnel and infrastructure to support Medexus’s anticipated future growth, the reduction in IXINITY net sales discussed above, and a $1.9 million increase in cost of goods sold related to previously disclosed failed batches during the IXINITY manufacturing process earlier in the fiscal year.
Net loss was $2.9 million for fiscal year 2022, compared to $28.3 million for fiscal year 2021.
Additional Information
Medexus’s financial statements and management’s discussion and analysis for the fiscal year ended March 31, 2022 are available on Medexus’s corporate website at www.medexus.com and in the company’s corporate filings on SEDAR at www.sedar.com.
Conference Call Details
Medexus will host a conference call at 8:00 AM Eastern Time on Thursday, June 23, 2022, to discuss the company’s operating and financial results and corporate updates for fiscal Q4 2022 and fiscal year 2022.
A replay of the call will be available approximately one hour following the end of the call through Thursday, June 30, 2022. To access the replay, please dial the following numbers:
877-481-4010 for Canadian and U.S. callers
+1 919-882-2331 for international callers
Conference ID: 45811
A replay of the webcast will be available on the Investors—News & Events—IR Calendar section of Medexus’s corporate website until Friday, June 23, 2023.
About Medexus
Medexus is a leader in innovative rare disease treatment solutions with a strong North American commercial platform and a portfolio of proven best-in-class products. Our current focus is on the therapeutic areas of hematology, auto-immune diseases, and allergy. We continue to build a highly differentiated company with a growing portfolio of innovative and high-value orphan and rare disease products that will underpin our growth for the next decade.
Our current leading products are Rasuvo™ and Metoject®, a unique formulation of methotrexate (auto-pen and pre-filled syringe) designed to treat rheumatoid arthritis and other auto-immune diseases; IXINITY®, an intravenous recombinant factor IX therapeutic for use in patients 12 years of age or older with Hemophilia B (a hereditary bleeding disorder characterized by a deficiency of clotting factor IX in the blood, which is necessary to control bleeding); and Rupall®, an innovative prescription allergy medication with a unique mode of action. We also hold exclusive US and Canadian rights to commercialize Gleolan™ (aminolevulinic acid hydrochloride or ALA HCl), an FDA-approved, orphan drug designated optical imaging agent currently indicated in patients with glioma (suspected World Health Organization Grades III or IV on preoperative imaging) as an adjunct for the visualization of malignant tissue during surgery.
We have also licensed treosulfan, part of a preparative regimen for allogeneic hematopoietic stem cell transplantation to be used in combination with fludarabine, for commercialization in the United States and Canada. Treosulfan was approved by Health Canada in June 2021 and is marketed in Canada as Trecondyv®. Treosulfan is currently the subject of a regulatory review process with the U.S. Food and Drug Administration.
Our mission is to provide the best healthcare products to healthcare professionals and patients. We strive to deliver on this mission by acting on our core values: Quality, Innovation, Customer Service, and Collaboration.
Contacts
For more information, please contact any of the following:
Ken d’Entremont, Chief Executive Officer
Medexus Pharmaceuticals Inc.
Tel: 905-676-0003
Email: ken.dentremont@medexus.com
Marcel Konrad, Chief Financial Officer
Medexus Pharmaceuticals Inc.
Tel: 312-548-3139
Email: marcel.konrad@medexus.com
Victoria Rutherford
Investor Relations Adelaide Capital
Tel: 1-480-625-5772
Email: victoria@adcap.ca
Forward-Looking Statements
Certain statements made in this press release contain forward-looking information within the meaning of applicable securities laws (forward-looking statements). The words “anticipates”, “believes”, “expects”, “will”, “plans”, “potential”, and similar words or expressions are often intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Specific forward-looking statements contained in this news release include, but are not limited to, statements regarding Medexus’s business strategy or outlook and future growth plans, expectations regarding future financial or operating performance (including with respect to the expected benefits of improvements made to the IXINITY manufacturing process and expected results from sales of Gleolan in the United States), ability to obtain FDA and other regulatory approvals for treosulfan and other product candidates, the timing of treosulfan launch in the United States, and competitive position of and anticipated trends and challenges in the company’s business and the markets in which it operates, among others. These statements are based on factors or assumptions that were applied in drawing a conclusion or making a forecast or projection, including assumptions based on historical trends, current conditions and expected future developments. Since forward-looking statements relate to future events and conditions, by their very nature they require making assumptions and involve inherent risks and uncertainties. Medexus cautions that although it is believed that the assumptions are reasonable in the circumstances, these risks and uncertainties give rise to the possibility that actual results may differ materially from the expectations set out in the forward-looking statements. Material risk factors include those set out in Medexus’s materials filed with the Canadian securities regulatory authorities from time to time, including Medexus’s most recent annual information form and management’s discussion and analysis; future capital requirements and dilution; intellectual property protection and infringement risks; competition (including potential for generic competition); reliance on key management personnel; Medexus’s ability to implement its business plan; Medexus’s ability to leverage its U.S. and Canadian infrastructure to promote additional growth; regulatory approval by relevant health authorities, including the FDA; product reimbursement by third party payers; litigation or expiry with respect to patents or other intellectual property rights; litigation risk; stock price volatility; government regulation; and potential third party claims. Given these risks, undue reliance should not be placed on these forward-looking statements, which are made only as of the date hereof. Other than as specifically required by law, Medexus undertakes no obligation to update any forward-looking statements to reflect new information, subsequent or otherwise.
Trademarks and trade names
This press release contains references to trademarks and service marks, including those belonging to other companies, persons, or entities. Solely for convenience, trademarks and trade names referred to in this document may appear without the “®” or “™” symbols. Each such reference should be read as though it appears with the relevant symbol. Any such references are not intended to indicate, in any way, that the holder or holders of the relevant intellectual property rights will not assert, to the fullest extent under applicable law, its rights to these trademarks and trade names.
Non-GAAP measures
Company management uses, and this press release refers to, financial measures that are not recognized under IFRS and do not have a standard meaning prescribed by generally accepted accounting principles (GAAP) in accordance with IFRS or other financial or accounting authorities (non-GAAP measures). These non-GAAP measures may include “non-GAAP financial measures” and “non-GAAP ratios” (each defined in National Instrument 52-112, Non-GAAP and Other Financial Measures Disclosure). Medexus’s method for calculating these measures may differ from methods used by other companies and therefore these measures are unlikely to be comparable to similarly-designated measures used or presented by other companies.
In particular, management uses Adjusted Net Income (Loss) and Adjusted EBITDA as measures of Medexus’s performance. Adjusted Net Income (Loss), EBITDA (earnings before interest, taxes, depreciation, and amortization) and Adjusted EBITDA are non-GAAP financial measures. In addition, Adjusted Net Income (Loss) may be presented on a per share basis.
An explanation and discussion of each of these non-GAAP measures, including their limitations, is set out under the heading “Preliminary Notes—Non-GAAP measures” in Medexus’s most recent management’s discussion and analysis. A reconciliation of each of these non-GAAP measures to the most directly comparable IFRS measure can be found under the heading “Reconciliation of Adjusted Net Income (Loss) and Adjusted EBITDA to Net Income (Loss)” below.
Reconciliation of Adjusted Net Income (Loss) and Adjusted EBITDA to Net Income (Loss) The following tables are derived from and should be read together with Medexus’s consolidated statement of operations for the three- and 12-month periods ended March 31, 2022. This supplementary disclosure is intended to more fully explain disclosures related to Adjusted Net Income (Loss) and Adjusted EBITDA and provides additional information related to Medexus’s operating performance. However, Medexus’s non-GAAP measures have limitations as analytical tools and should not be considered in isolation or as a substitute for analysis of Medexus’s financial information as reported under IFRS.